In the broad and shifting field of medicine, there is an understanding that progress is both necessary and inevitable. Healthcare providers have more access to knowledge than ever before, and this knowledge is being constantly updated and refined by experts all over the globe. As more research is done, and more studies are circulated, medical professionals are faced with an ever-growing fountain of evidence. As incredible as this vast body of knowledge is, it speaks to a specific problem that needs to be addressed - its vastness.
For example, the appropriate prescription of antibiotics is a complicated, multistep decision-making process that involves consideration of the latest evidence from many disciplines including microbiology, pharmacology, epidemiology, and more. This can be further complicated by resource availability, patient history and competing priorities for treatment. Many prescribers are already overburdened by a lack of resources and an excess of patients or lengthy working hours, and cannot be expected to carry the weight of this process alone. It is more important than ever that we support these providers at the point of care, and make the process of medical treatments easier for both patients and practitioners.
Dr. Mike Sharland is the current chair of the Antibiotic Working Group for the The World Health Organization’s Essential Medicines List (WHO EML), and has experienced firsthand the need for quick and early access to medical information. A pediatric infectious disease specialist for 40 years, Dr. Sharland began his journey working with children suffering from a range of illnesses, from HIV to sepsis to cancer. “We noticed a lot of multidrug-resistant bacterial infections that were getting harder to treat, and less sensitive to antibiotics”, he says. Dr. Sharland is based in London, UK, but brings up a problem that affects communities globally and can be particularly hard-hitting for low and middle-income countries; the increasing prevalence of antimicrobial resistant infections, largely driven by the inappropriate use of antimicrobials. By the time more appropriate treatment, or a more knowledgeable health authority can be utilized, it’s too late for many patients. Access to the best, most up-to-date treatment information and early intervention are the most important factors when it comes to combating antimicrobial resistance (AMR).
Introducing the Essential Medicines List
The solution put forth by Dr. Sharland, as well as other researchers in the field of infectious diseases, is to encourage and implement the use of the WHO EML. The EML was created as a standardized guide for countries to refine based on local treatment guidance and priorities. It also endeavours to improve equitable access to medications across different countries and support more appropriate prescribing. When it was originally released in 1977, 240 essential medicines were included, of which 16 were antibiotics. Now, the 2021 WHO EML includes 479 medicines and 39 antibiotics.

Dr. Mark Loeb, professor of Pathology and Molecular Medicine at McMaster University and a member of the WHO EML Working Group reiterated the importance of not only access to the EML, but also a guide dedicated to the management of common infections. The EML is a large volume, with an ever-growing list of medicines under many different specialties and for many different conditions. Dr. Loeb says he noticed in 2017 that the antibiotic list within the EML hadn’t been updated for many years, and the WHO lacked an empirical, evidence-based system to both classify and justify the antibiotics on the list.
Developing the EML AWaRe Antibiotic Book
“It started out as a brainstorming session [in the WHO EML Working Group] about what the way forward would be,” he explains. “We needed to generate evidence to support what countries should be using for empiric antibiotics.” The EML AWaRe Antibiotic Book was conceived to provide evidence-based guidance for the management of the 35 most common infections in adults and children in both the primary healthcare and hospital settings. Dr. Loeb’s pivotal role was to generate this evidence for the AWaRe classifications, a system to which he was integral in developing, as well as creating a book to package and distribute this information. “The essence of this book is that it’s the EML of antibiotics,” he explains.
Most prescribers of antibiotics are not infectious disease experts, and thus may not be able to independently integrate the latest research into their already complex decision making process. Instead, The EML Working Group recommends understanding and utilizing the AWaRe system, which classifies antimicrobials into three groups:
Access: Narrow spectrum, generally less expensive and safer with a lower resistance potential.
Watch: Broader spectrum, generally more expensive, with a higher potential to select for resistance.
Reserve: ‘Last-resort’ antibiotics for use against multidrug-resistant bacteria.
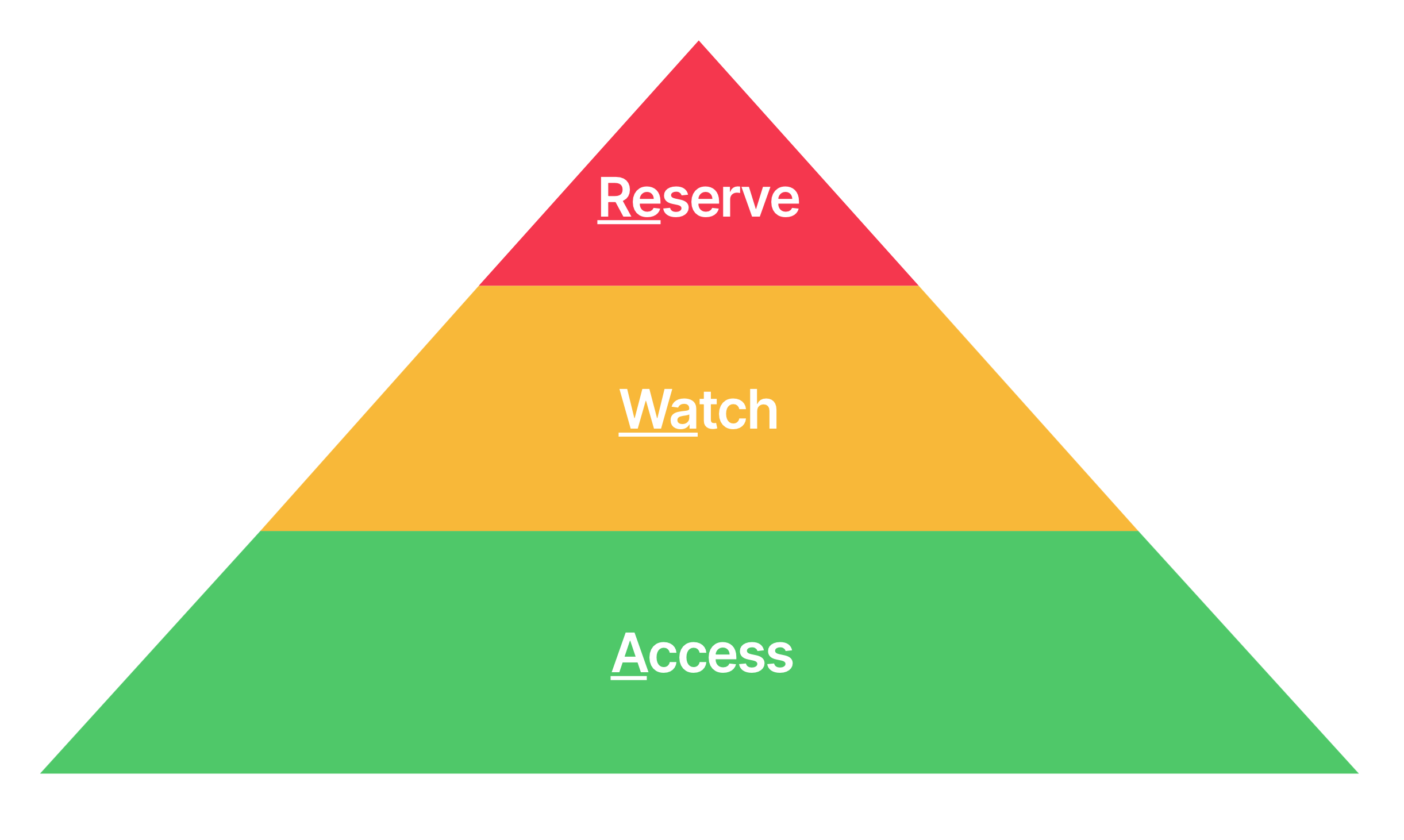
This classification framework, combined with use of the EML AWaRe Antibiotic Book, yields the best results when prescribing.
Most researchers agree that a standardized prescribing guide for empiric antibiotics is a necessary resource that can supplement local protocols and help countries improve the appropriateness of prescribing. “There is a very high usage of oral watch antibiotics in high, middle and low-income settings. This is not just a low and middle-income setting issue”, Dr. Sharland pointed out. The EML AWaRe Antibiotic Book puts a strong emphasis on symptomatic care for mild infections in otherwise healthy patients and encourages the use of access antibiotics and the judicious use of watch antibiotics.
The next important step for the EML AWaRe Antibiotic Book, according to both Dr. Sharland and Dr. Loeb, is encouraging widespread international use in countries and regions that are most in need of treatment guidance for infectious diseases. This means distilling medical information into a digestible and usable format, and working with local health authorities to encourage distribution of this information. Prescribers in different regions may have more or less access to certain antibiotics, or local susceptibility patterns may affect their first line of treatment, and all of this needs to be taken into account when implementing guidelines.
Dr. Sharland and Dr. Loeb also stress the importance of digitization in the future of this implementation process. Digitizing the EML AWaRe antibiotic book onto one streamlined platform, such as Firstline, will make the guidance easy to adopt as well as easily adaptable to fit the local needs of health organizations all over the world, improving the experience of prescribers and patients alike.
Despite differences in prescribing practices and access to medicines, the effects of antimicrobial resistance can be seen in communities across the world. “WHO’s guidance for managing common infectious diseases is just the beginning of the journey,” says Dr. Sharland. “There needs to be a strong educational framework around this, thinking about what the implications are for policy, and how this can lead into implementation at the country level, regional level, [and] hospital level.”
Learn more about the WHO EML (22nd List, 2021), and the WHO EML AWaRe Antibiotic Book.
